Molecule Shapes
|
|
|
Back to HTML5 Version |
About
Topics
- Molecules
- VSEPR
- Bonds
- Lone Pairs
Description
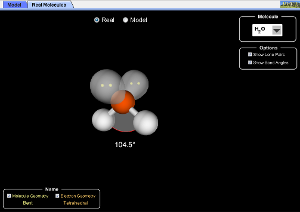
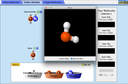
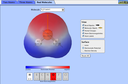
Explore molecule shapes by building molecules in 3D! How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules!
Sample Learning Goals
- Recognize that molecule geometry is due to repulsions between electron groups.
- Recognize the difference between electron and molecular geometry.
- Name molecule and electron geometries for molecules with up to six electron groups surrounding a central atom.
- Compare bond angle predictions from the VSEPR-based model to real molecules.
- Describe how lone pairs affect bond angles in real molecules.
Keywords
For Teachers
Teacher Tips
| Overview of sim controls, model simplifications, and insights into student thinking ( PDF ). |
Teacher-Submitted Activities
Translations
Related Simulations
Software Requirements
| Windows | Macintosh | Linux |
|---|---|---|
|
Microsoft Windows XP/Vista/7 Sun Java 1.5.0_15 or later
|
OS 10.5 or later Sun Java 1.5.0_19 or later
|
Sun Java 1.5.0_15 or later
|
Credits
| Design Team | Third-party Libraries | Thanks To |
|---|---|---|
|
|